Treatment of inhibitors
The development of anti-factor VIII (FVIII) antibodies (inhibitors) is the most serious treatment-related complication in patients with hemophilia A, rendering standard replacement therapy ineffective, heightening the risk for uncontrollable bleeding and morbidity, decreasing quality of life, and increasing healthcare costs.
Current management includes bypassing agents to treat and prevent bleeding and immune tolerance induction (ITI) to attempt inhibitor eradication. For those patients with low responding inhibitors treatment with high doses of FVIII is also an alternative.

Bypassing agents, aPCC and rFVII
-
aPCC is a pooled human plasma derivative that contains factors II, IX and X mainly in non-activated form as well as activated factor VII with approved indications* for:
- Treatment , prophylaxis and surgical interventions of bleeding in haemophilia A and B patients with Factor VIII and FIX inhibitor.
- Treatment and prophylaxis of bleeding in non haemophiliacs with acquired inhibitors to Factors VIII, IX, XI.
- Other uses include patients with an inhibitor to von-Willebrand Factor, and the used in combination with Factor VIII concentrate for long-term therapy to avoid breakthrough bleeding during ITI.
-
rFVII Eptacog alfa (activated) is recombinant coagulation factor VIIa (rFVIIa). Indicated * for the treatment of bleeding episodes and for the prevention of bleeding in those undergoing surgery or invasive procedures in the following patient groups:
- In patients with congenital haemophilia with inhibitors to coagulation factors VIII or IX > 5 Bethesda Units (BU).
- In patients with congenital haemophilia who are expected to have a high anamnestic response to factor VIII or factor IX administration.
- In patients with acquired haemophilia.
- In patients with congenital FVII deficiency.
- In patients with Glanzmann's thrombasthenia with antibodies to GP IIb - IIIa and/or HLA, and with past or present refractoriness to platelet transfusions.

Immune Tolerance Induction: Eradication of Inhibitor
- Immune tolerance induction (ITI) is the use of frequent exposure to FVIII until antigen acceptance is achieved and normal replacement FVIII kinetics are restored.
- The goal of ITI is the permanent eradication of the inhibitor and the restoration of the clinical and biologic efficacy of FVIII concentrates.
- The major complication experienced by patients with inhibitors is the difficulty to control bleeding, which often can be managed with bypass agents while waiting for immune tolerance to eradicate the inhibitor.

Immune tolerance induction –40 years of experience
- Experience has shown success rates of between 60% and 80% for patients with hemophilia A administered ITI 1-3
- Inhibitor eradication is required to ensure a more normal quality of life and reduced long-term morbidity.

Classic Immune tolerance protocols
- Summary of the main classic immune tolerance induction protocols 5

Actual treatments will vary from center to center and most protocols have been based on these classic protocols.

Predictors for ITI Success
- There have been parameters identified retrospectively that can influence both time to successful ITI and success rate in severe hemophilia A patients with high titer inhibitors 6

Adapted from Di Michele DM. 6

Effect of Product Type on ITI Success
Despite more than 35 years of clinical experience, the optimal ITI regimen, including the choice of FVIII product for immune tolerance, has not been established largely due to the lack of evidence from large-scale and methodologically rigorous trials 7
Several studies have reported success in patients with poor prognostic factors receiving ITI treatment with both pd FVIII/ VWF concentrates and rFVIII 8-20
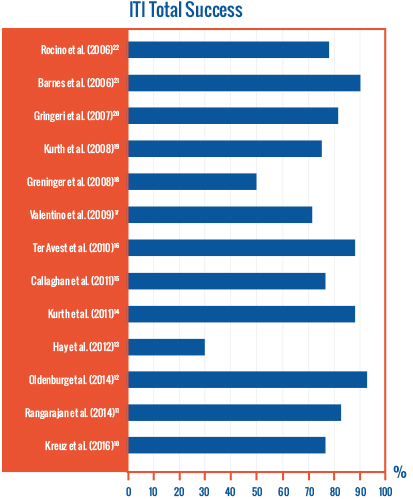

The role of VWF in immune tolerance induction with pdFVIII/FVW concentrates
-
There are hypothetical mechanisms that have been proposed to explain why the inclusion of VWF in FVIII concentrates may increase the efficacy of ITI: 21-22
- FVIII/VWF complex may block anti-FVIII-antibody binding to epitopes located in the VWF-binding A3 and C2 domains of the FVIII light chains.
- A prolonged FVIII presentation to the immune system.
- Immunomodulatory effect of VWF.
-
There are several in vitro and in vivo observations in published studies:
- The lower inhibitory activity against FVIII/FVW complex compared to recombinant FVIII in inhibitor plasmas with anti-C2-specifity 21-25
- Higher recovery when the infused FVIII concentrate contained VWF during treatment of patients with hemophilia A and an inhibitor against the light chain of FVIII 23-26
"Native pdFVIII/VWF complex has lower sensitivity to FVIII inhibitors than the combination of isolated FVIII and VWF proteins. Impact on Bethesda assay titration of FVIII inhibitors." Bravo MI. Haemophilia 2014 27
- Objective: To test inhibitor reactivity in different combinations of VWF, FVIII concentrates (plasma-derived and recombinant) and inhibitors.
-
Results:
- Inhibitor titers for plasma and pdFVIII/VWF complex were comparable at all time points.
- Inhibitor titers for all concentrates of isolated FVIII were significantly higher than those for plasma or pdFVIII/VWF complex (1.4–1.9 fold) even after pre-incubation with VWF.
"Beneficial effect of VWF in FVIII recovery of different FVIII concentrates in a mouse model of severe hemophilia with high inhibitor titers" Bravo MI. J Thromb Haemost., 2015 28
- Objective: To evaluate in a mouse model of severe hemophilia the differences of FVIII in vivo recovery between different FVIII concentrates –with and without VWF– in the presence of high inhibitor levels (>5BU).
-
Results:
- In the presence of high titer inhibitors FVIII recovery was higher for native pdFVIII/VWF when compared to concentrates of isolated FVIII (20.8% vs. 5.4% for rFVIII and 1.2% for pdFVIII, respectively).
- Similar results were achieved with low titer inhibitors: 71.1% vs. 31.4% for rFVIII and 25% for pdFVIII.


Economics of ITI
Treating hemophilia patients with inhibitors is costly regardless of the preferred treatment approach. In particular, although ITI treatment is associated with a high cost, the overall lifetime treatment costs are substantially lower than treating prophylactically with bypassing agents 29
A cost analysis on factor VIII alloantibody inhibitors on immune tolerance induction vs. prophylaxis and on-demand with bypass treatment was recently published: 29
- Compared to on-demand and prophylactic treatment with bypassing agents, ITI was found to be less expensive when considering lifetime costs.
- Even though on demand treatment was not substantially more costly, it carries much more risk due to the occurrence of four times as many bleeding events.
- As a result, ITI may be associated with lower lifetime costs than when treating on-demand or prophylactically with bypassing agents. The economic benefit of treating with ITI is best achieved the earlier one begins immune tolerance therapy in a patient with an inhibitor.
It is important to note that treatment varies by patient. Be sure to consult a healthcare professional before making any treatment decisions and continue to remain informed to help facilitate care.
It is important to note that treatment varies by patient. Be sure to consult a healthcare professional before making any treatment decisions and continue to remain informed to help facilitate care.
REFERENCES
- Coppola A., Di Minno MN. , Santagostino E. Optimizing management of immune tolerance induction in patients with severe haemophilia A and inhibitors: towards evidence-based approaches. Br J Haematol. 2010; 150:515–28.
- Brackmann H. et al. Immune tolerance for the treatment of FVIII inhibitors - Twenty years "Bonn protocol". Vox Sang 1996; 70 (Suppl. 1):30-35.
- Dimichele D. The North American Immune Tolerance Registry: contributions to the thirty-year experience with immune tolerance therapy. Haemophilia 2009; 15:320–8.
- Mariani G, Kroner B. Immune Tolerance Study Group (ITSG). Immune tolerance in hemophilia with factor VIII inhibitors: predictors of success. Haematologica 2001; 86:1186–93.
- Benson G. et al. Immune tolerance induction in patients with severe hemophilia with inhibitors: expert panel views and recommendations for clinical practice. Eur J Haematol 2012 May; 88(5):371-9.
- Di Michele DM. Immune tolerance induction in haemophilia: evidence and the way forward. J.Thromb Haemost. 2011 Jul;9 Suppl 1:216-25.
- Di Minno G., Coppola A. A role for von Willebrand factor in immune tolerance induction in patients with haemophilia A and inhibitors? Blood Transfus. 2011 May; 9(Suppl 2): s14–s20.
- Kreuz W. et al. First prospective report on immune tolerance in poor risk haemophilia A inhibitor patients with a single factor VIII/von Willebrand factor concentrate in an observational immune tolerance induction study. Haemophilia 2016 Jan; 22(1):87-95.
- Rangarajan S. et al. Adult hemophilia A patients with inhibitors: successful ITI with a single FVIII/VWF product. Haemophilia 2014 Nov; 20(6):e414-7.
- Oldenburg J. et al. Primary and rescue ITI in children and adults: a multicenter international study with a VWF-containing plasma-derived FVIII concentrate". Haemophilia 2014 Jan; 20(1):83-91.
- Hay CR. et al. The principal results of the International Immune Tolerance Study: a randomized dose comparison. Blood 2012 Feb 9; 119(6):1335-44.
- Kurth M. et al. The use of a single von Willebrand factor-containing, plasma-derived FVIII product in hemophilia A immune tolerance induction: the US experience. J Thromb Haemost. 2011 Nov; 9(11):2229-34.
- Callaghan MU. et al. Immune tolerance induction in 31 children with haemophilia A: is ITI less successful in African Americans? Haemophilia 2011 May; 17(3):483-9.
- Ter Avest PC. et al Successful low dose immune tolerance induction in severe haemophilia A with inhibitors below 40 Bethesda Units. Haemophilia 2010 May;16(102):71-9.
- Valentino LA. et al. Experience with a third generation recombinant factor VIII concentrate for immune tolerance induction in patients with haemophilia A. Haemophilia 2009 May; 15(3):718-26.
- Grenninger DA. et al. The use of factor VIII/von Willebrand factor concentrate for immune tolerance induction in haemophilia A patients with high-titer inhibitors: association of clinical outcome with inhibitor epitope profile. Haemophilia 2008 Mar;14(2):295-302.
- Kurth M.A. et al. Immune tolerance therapy utilizing factor VIII/von Willebrand factor concentrate in haemophilia A patients with high titer factor VIII inhibitors. Haemophilia 2008 Jan;14(1):50-5.
- Gringeri A. et al. Immune tolerance induction with a high purity von Willebrand factor/VIII complex concentrate in haemophilia A patients with inhibitors at high risk of a poor response. Haemophilia 2007 Jul; 13(4):373-9.
- Barnes C. et al. Canadian multi-institutional survey of immune tolerance therapy (ITT) -- experience with the use of recombinant factor VIII for ITT. Haemophilia 2006 Jan; 12(1):1-6.
- Rocino A. et al. "Immune tolerance induction with recombinant factor VIII in hemophilia A patients with high responding inhibitors" Haematologica 2006 Apr;91(4):558-61.
- Kallas A. and Talpsep T. von Willebrand factor in factor VIII concentrates protects against neutralization by factor VIII antibodies of haemophilia A patients. Haemophilia 2001 Jul;7(4):375-80.
- Astermark J. et al. Impact of inhibitor epitope profile on the neutralizing effect against plasma-derived and recombinant factor VIII concentrates in vitro. Haemophilia 2003 Sep; 9(5):567-72.
- Berntorp et al. Variation in factor VIII inhibitor reactivity with different commercial factor VIII preparations. Haemophilia 1996 Apr; 2(2):95-9.
- Suzuki T. et al. Factor VIII inhibitor antibodies with C2 domain specificity are less inhibitory to factor VIII complexed with von Willebrand factor. Thromb Haemost. 1996 Nov; 76(5):749-54.
- Gensana M. et al. Influence of von Willebrand factor on the reactivity of human factor VIII inhibitors with factor VIII. Haemophilia 2001 Jul; 7(4):369-74.
- Inoue T. et al. Higher recovery of factor VIII (FVIII) with intermediate FVIII/von Willebrand factor concentrate than with recombinant FVIII in a haemophilia A patient with an inhibitor. Haemophilia 2006 Jan; 12(1):110-3.
- Bravo M.I. et al. Native plasma-derived FVIII/VWF complex has lower sensitivity to FVIII inhibitors than the combination of isolated FVIII and VWF proteins. Impact on Bethesda assay titration of FVIII inhibitors." Haemophilia 2014 November; 20(6): 905–911.
- Bravo M.I. et al. Beneficial effect of VWF in FVIII recovery of different FVIII concentrates in a mouse model of severe hemophilia with high inhibitor titers Thromb Haemost., June 2015, Volume 13, Issue Supplement S2, Pages 826-827.
- Earnshaw SR. et al. Factor VIII alloantibody inhibitors: cost analysis of immune tolerance induction vs. prophylaxis and on-demand with bypass treatment. Haemophilia 2015 May; 21(3):310-9.

